Sapien receives DPIIT recognition
/0 Comments/in Blog/by adminPleased to get the certificate from #DPIIT. Heartfelt thanks to @Saravjeet Chahal who helped resolve the glitches at #StartUp India website due to CIN number mismatch.
Attention all Telangana startups that were formed in combined AP, before formation of Telangana, like #SapienBio, if you are facing persistent problems getting your application completed on #StartupIndia website, it may be due to your CIN number with AP not getting validated by MCA, and AP getting morphed to TG, which then creates other problems. Suggest contacting Saravjeet who will help. The tollfree call numbers did not. Thank you #nitiayog and @Sanjana Mohanty, @Aastha Grover for helping resolve our 8 month long quest for recognition.
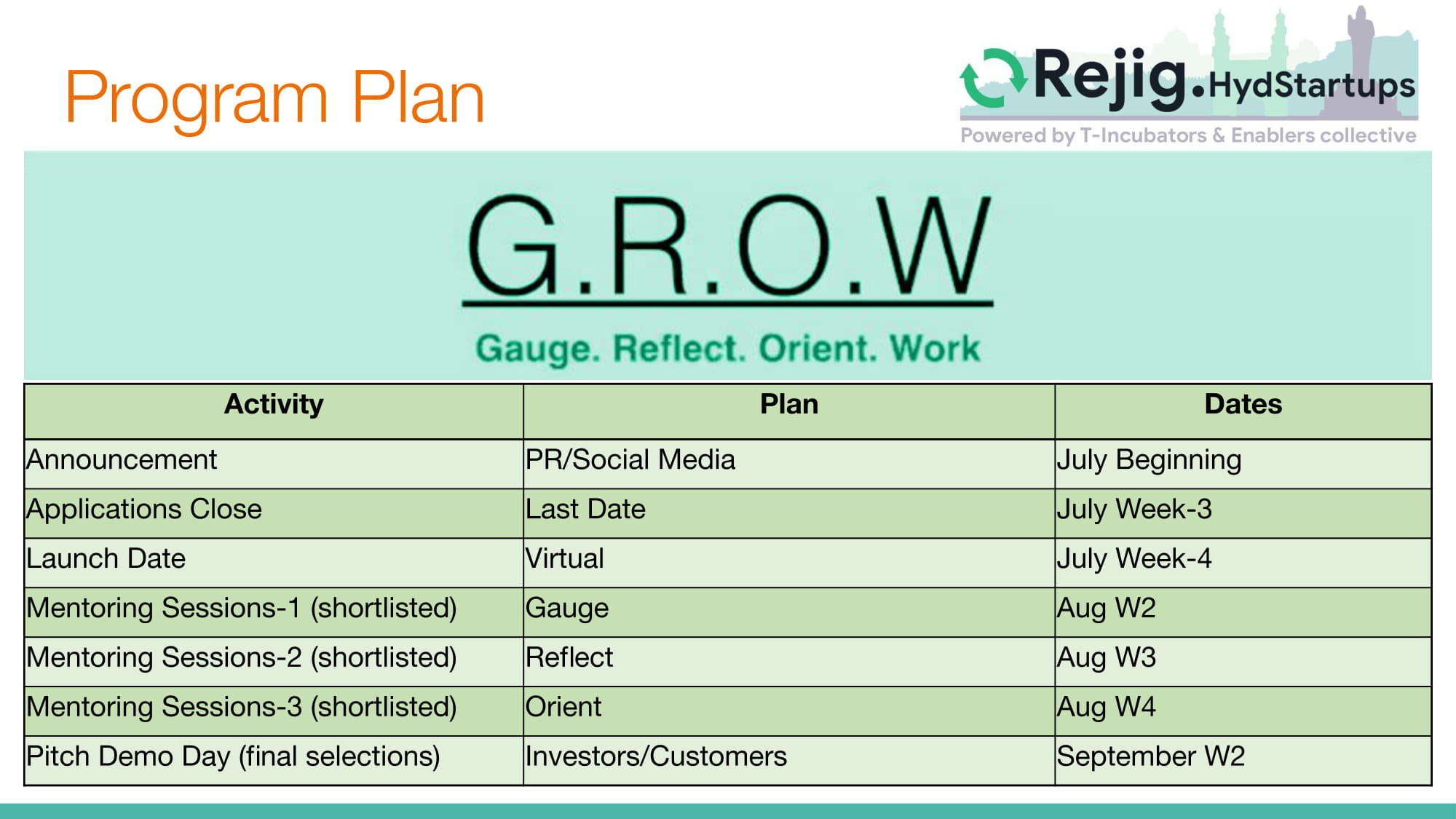
Rejig.HydStartups kick-off G.R.O.W
/0 Comments/in Blog/by admin
First of the Rejig.Hyderabad marathon sessions happened yesterday evening. A total of 13 companies, broadly in area of healthcare, were given 3 minutes each to pitch virtually in front of 4 mentors: @Ambuj Chaturvedi, @Vijay Bhaskar, @Neetika Maheshwari, @Vivek Bhargava, with @Paridhi Gupta and @Ramjee Pallela diligently keeping us all on time. It was heartening to get instant mentor feedback to our pitch for growth funds in that they all said Sapien had clearly proven traction and market validation for its services and products. They were keen to see our roadmap for scale-up, product and IP potential, and interested in seeing how the structured patient data we have built up carefully for breast, lung, renal, colorectal, gliomas etc. could be made available for research at universities to build data analytics & AI capabilities. I look forward to the 1-on-1 mentoring sessions to help refine our pitch for success.
To contact us – please write to queries@sapienbio.com
Sapien Biosciences selected for Rejig.HydStartups Corporate Market Access and Investor Program
/0 Comments/in Blog/by admin
Rejig.HydStartups has selected 100 start-ups for programs that help them reposition their business models post-pandemic. Sapien Biosciences is one of the lucky companies selected for the Corporate Market Access and Investors Program.
Anti-cancer screening of herbal drugs using cancer patient cells
/0 Comments/in Blog/by admin[fliph5 id=”tszft-qjeo” width=”600px” height=”350px”]
Our CEO, Dr. Jugnu Jain, wins Niti-Aaayog’s Women Transforming India Award on International Women’s Day, March 2020.
/0 Comments/in Blog/by adminhttps://www.youtube.com/watch?v=Grg30L36B50

The Fourth Edition of the WTI Awards built on last year’s theme of ‘Women and Entrepreneurship’ was announced in August 2019. NITI Aayog received over 2300 applications from across the country in November, 2019. On 8th March 2020, the Top 15 women entrepreneurs were awarded at the Women Transforming India Awards ceremony held at Sushma Swaraj Bhawan in New Delhi.
Sapien’s breast cancer database showcased @BioAsia2020, shortlisted by an expert selection committee for Startup Stage @ BioAsia 2020.
/0 Comments/in Blog/by Sapienbio Staff
Sapien Biosciences team @ BioAsia 2020, at Stall # S14. Sapien was selected from the Startup Stage competition and awarded free exhibit space and delegate pass at BioAsia. Here we showcased several breast cancer patients’ tissue-derived in vitro models for testing new drugs and markers at Sapien. The deep database of ~10,000 Indian breast cancer patients’ data was also showcased. These data can be used for developing Artificial Intelligence-based algorithms to improve the diagnosis, treatment and survival of future breast cancer patients.
Lung Cancer 2018
/0 Comments/in Blog/by adminDr Jain of Sapien selected to participate at Global Entrepreneurs Summit 2019 at the Hague, NL
/0 Comments/in Blog/by adminDr. Jugnu Jain, Co-Founder and CEO of Sapien Biosciences, has been selected to participate in the Global Entrepreneur Summit (GES), 3-5th June 2019, at the Hague in Netherlands. Our biobank will represent India in the area of Health at the summit. We have also been invited to the select discussion with the U.S. Department of Health and Human Services Deputy Secretary Eric D. Hargan and CEO of Royal Philips, Frans van Houten, on Health Policy and Innovation.
Sapien will be showcased in front of a select group of decision makers, including funders, founders, experts and policymakers. Leading American, European, Asian and global companies alongside institutional investors such as APG and PGGM have committed to take part in the GES.
As GES website states – it gathers governments, the private sector and a powerful partner network to turn challenges into business opportunities worldwide. It is co-hosted by two of the world’s most innovative countries, the United States and the Netherlands.
2019 is the 9th edition of GES, which will focus on global opportunities for the next wave of entrepreneur-led innovation in scalable enterprises. We have reached out to other Healthpreneurs at the summit and look forward to meeting them.

Locate Us
Sapien Biosciences,
1st Floor, AIMSR Building,
Apollo Health City,
Jubilee Hills,
Hyderabad 500 096
Phone:
+91-99859 25652
+91-73863 01984
Email:
info@sapienbio.com
queries@sapienbio.com







Recent Comments